The first major update to OC4 went live early today, with new features that extend your study’s flexibility so much that we wanted to call them out in this post.
“Common Events”: the new, intuitive way to accommodate AEs and conmed changes
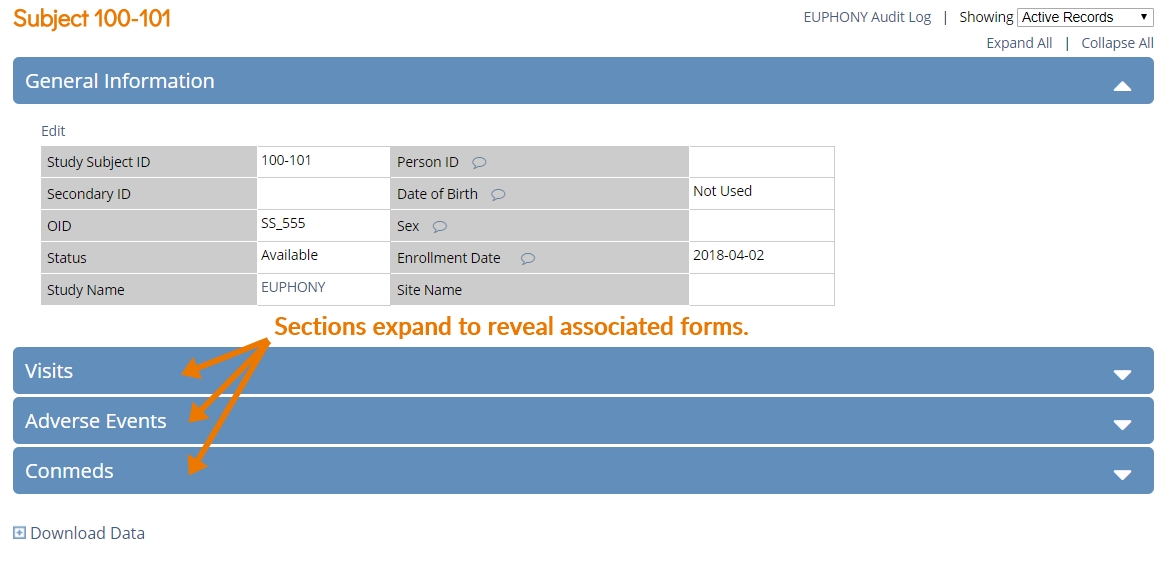
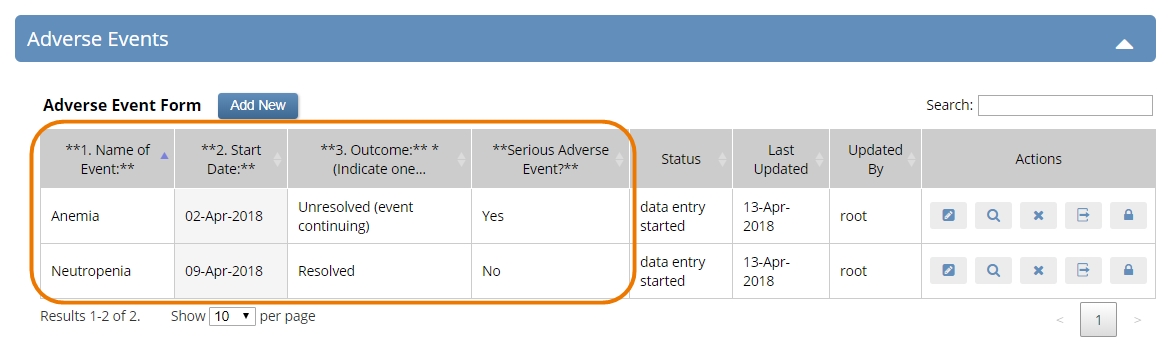
Common events are not associated with a visit date, and do not occur at a scheduled time-point. Instead, they are incidental events that often recur, such as adverse events and concomitant medications. This update makes it easy to include common events in your study. You can include multiple forms in a common event, which allows each form to repeat independently within the event.
Study designers can even select key fields in the forms to display those field values on the (new, UX-optimized) Subject Details page, allowing users to view form data without having to open the form.
“Upload a file, any file”
You now have several distinct widgets for collecting files.
Image, audio, and video widgets allow users to upload these file types, which may then be viewed or played within the browser window.
We have also added a generic file widget for uploads of any type. (However, these files do not support a preview in the browser window.)
Any uploaded file may be downloaded from within the form.
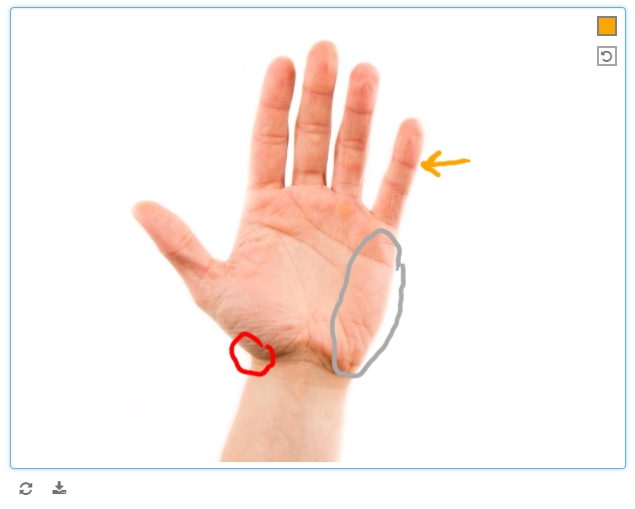
Draw, annotate, or sign
Used judiciously, free text fields can enrich the data collected in a study. So just image what free drawing can do. This latest update supports:
- Drawing on a blank canvas, from a palette of 12 colors plus black, with a mouse, finger, or stylus
- Annotating an uploaded image with freehand notes or diagrams (from the same color palette, using a mouse, finger, or stylus)
- Signing one’s name, in black only, with a mouse, finger, or stylus
The annotate and draw widgets allow the user to undo individually added lines.
Central User Management
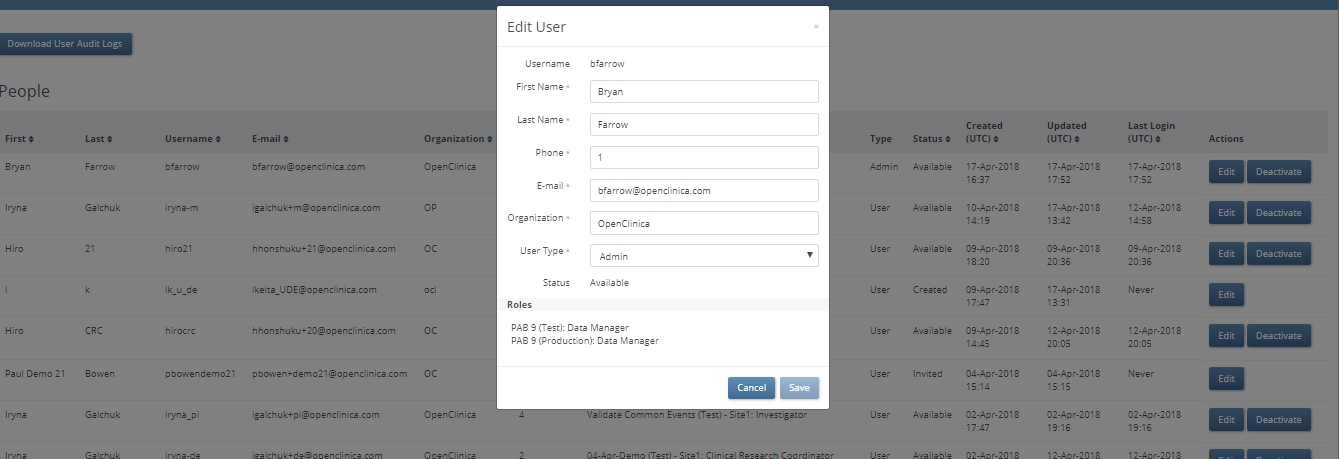
A new feature allows administrators to view a directory of all users created for at least one test or production environment in their instance. Each user record indicates:
- First name
- Last name
- Username
- Email address
- Organization (e.g. Site 1234)
- all roles for all study environments (e.g. EUPHONY Study (Test): Clinical Research Coordinator, PRAXIS Study (Production): Data Entry Person, etc.)
- Role type (Admin or User)
- Status (Created > Invited > Available/Signed In)
- Date and time created
- Date and time last updated
- Date and time of last login
An edit button allows an administrator to update a user’s first name, last name, phone number, e-mail address, organization, or user type (i.e., attributes that are independent of any one study or environment). A deactivate button removes access for that user to any study environment to which they had been assigned. For deactivated users, a Reactivate button restores all access.